/the-periodic-table--digital-illustration--73016803-598b218ec41244001024af78.jpg)
The alkaline earth metals can be found in. 1, reacts readily with metals to form compounds that can be broadly classified as salts and, therefore, are known as the halogens, which is derived from a combination of Greek words that translate to 'salt makers. Alkalis are often hydroxides of alkali metals (metals that belong to group IA of the periodic table, including Li, Na, K, Rb, Cs, and Fr). Alkaline earth metals are sometimes called group 2 elements because they are located in the second group or column of the periodic table of elements. Each of the elements found in this column, which are boxed in lavender in Figure 2.2.1 2.2. Although hydrogen is in group 1 (and also in group 17), it is a nonmetal and deserves separate consideration later in this chapter. Definition of alkali any compound that has highly basic properties. 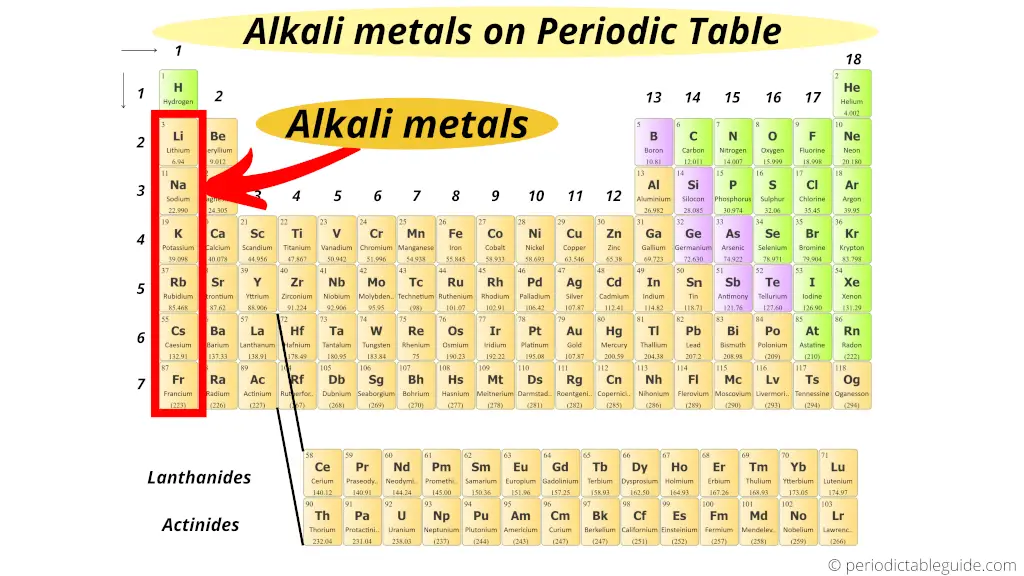
The alkali metals lithium, sodium, potassium, rubidium, cesium, and francium constitute group 1 of the periodic table. The group 1 metals are lithium, sodium, potassium, rubidium, caesium and francium and they are found in the first column of the periodic table. They form alkaline solutions when they react with water. Although beryllium, cadmium, mercury, and lead are readily available, there are limitations in their use because of their toxicity. The group 1 metals are known as the alkali metals. Elemental magnesium, aluminum, zinc, and tin are important in the fabrication of many familiar items, including wire, cookware, foil, and many household and personal objects. The coating is a nonreactive film of oxide or some other compound. This page introduces the Alkali Metals in Group 1 of the Periodic Table. The formation of this protective coating is passivation. Alkali metals are any of the elements found in Group IA of the periodic table (the first column). Although hydrogen is in group 1 (and also in group 17), it is a nonmetal and deserves separate consideration later in this chapter. Part of the reason why these elements react slowly is that these elements react with air to form a protective coating. The alkali metals lithium, sodium, potassium, rubidium, cesium, and francium constitute group 1 of the periodic table. 
However, it is possible to isolate elemental beryllium, magnesium, zinc, cadmium, mercury, aluminum, tin, and lead from their naturally occurring minerals and use them because they react very slowly with air. Most of the representative metals do not occur naturally in an uncombined state because they readily react with water and oxygen in the air. A salt is an ionic compound consisting of cations and anions.Ī salt is an ionic compound consisting of cations and anions. Each box represents an element and contains its atomic number, symbol, average atomic mass, and (sometimes) name. 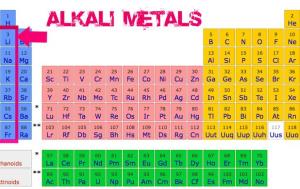
In general, the combination of a metal and a nonmetal produces a salt. A modern periodic table arranges the elements in increasing order of their atomic numbers and groups atoms with similar properties in the same vertical column ( Figure 5.3 ). Unlike metals, which typically form cations and ionic compounds (containing ionic bonds), nonmetals tend to form anions or molecular compounds. The remaining representative elements are nonmetals. Alkalies are strong bases that turn litmus paper from red to blue they react with acids to yield neutral salts and they are caustic and in concentrated form are corrosive to organic tissues. A metalloid is an element that has properties that are between those of metals and nonmetals these elements are typically semiconductors. alkali, any of the soluble hydroxides of the alkali metals i.e., lithium, sodium, potassium, rubidium, and cesium. This group includes the elements beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra). In addition to the representative metals, some of the representative elements are metalloids. Alkaline earth metals are the elements that belong to group 2 of the periodic table i.e., the second vertical column. Nonmetals are shown in green, metalloids in purple, and the transition metals and inner transition metals in blue. The rock salt structure is by Solid State - Own work, Public Domain, /w/inde.?curid=3837309 the CsCl structure image by Solid State - Own work, Public Domain, /w/inde.?curid=3837330.\): The location of the representative metals is shown in the periodic table. (B) CsCl structure formed by CsCl, CsBr, and CsI showing square coordination of eight halides about each Cs atom. This is the reason why the alkali metals are regarded as the most electropositive elements (with caesium and francium being the most electropositive elements in the entire periodic table). Review the Periodic Table of the Elements in other formats in Appendix A (credit: Chemistry (OpenStax), CC BY 4.0). (A) Rock salt structure formed by LiX, NaX, KX, and RbX (X = F, Cl, Br, and I) showing octahedral coordination of six halides about each metal ion. Figure 3.2g: The periodic table organizes elements with similar properties into groups.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
